The presentation is designed to be accessible to anyone with intermediate undergraduate mathematics, physics and chemistry.ĭieser Download kann aus rechtlichen Gründen nur mit Rechnungsadresse in A, D ausgeliefert werden.
In presenting a broad overview of the of the physical foundations of dispersion forces, the book provides theoretical, physical and synthetic chemists, as well as solid-state physicists, with a systematic understanding of the origins and consequences of these ubiquitous interactions. 13 Furthermore, as London dispersion forces result from a quantum-mechanical.

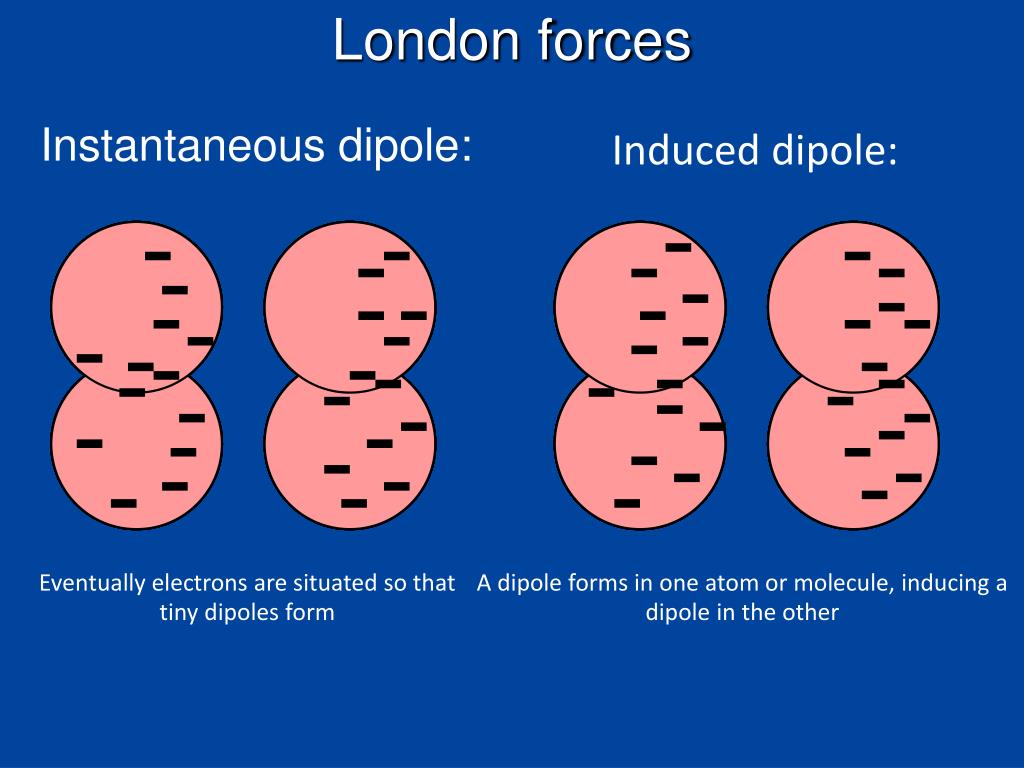
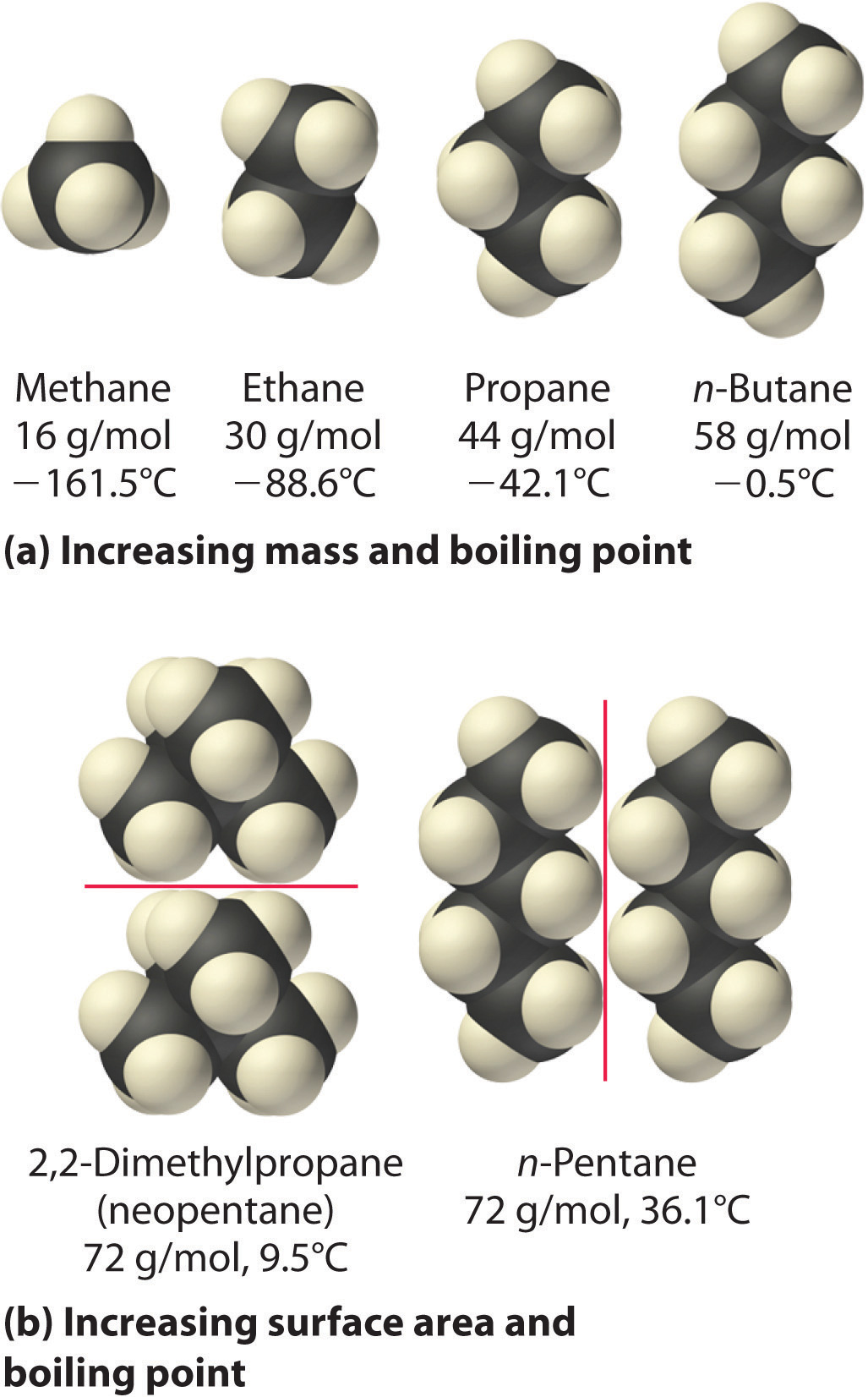
They can then attract to each other in a. These are molecules where the electronegativity difference between the. London dispersion forces exist in non polar molecules. They are very often found in non polar molecules that are in simple covalent compounds or elements. 35 This view is further strengthened by the notion that solvation negates any steric attraction between solvated molecules, thus making it irrelevant for chemistry occurring in solution. This instantaneous dipole can induce a dipole in another nearby non-polar molecule. London dispersion forces are the weakest type of intermolecular forces and require very little energy to overcome them. London dispersion forces result from the interaction between the electrons of one molecule and the nuclei of another. Individual London dispersion forces are often very weak, which has led to it commonly being regarded as an insignificant force. It covers a wide range of system, from small intermolecular complexes, to organic molecules and crystalline solids, through to biological macromolecules and nanostructures. London Dispersion Forces: The weakest intermolecular force. This book is an overview of current understanding of the physical origin and modelling of London dispersion forces manifested at an atomic level. Recent years have seen the development of new, physically well-founded models, and dispersion-corrected density functional theory (DFT) is now a hot topic of research. London dispersion interactions are responsible for numerous phenomena in physics, chemistry and biology.

